|
9/8/2023 0 Comments What does entropy mean 
In both cases the energy is converted into heat by the plants. 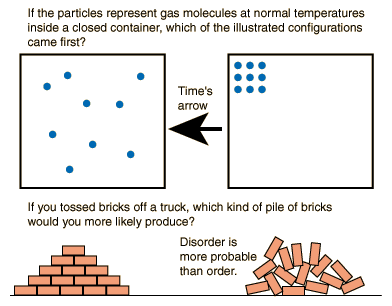
That means plants use the low entropy of sunlight to get low entropy for themselves? That’s pretty clever.Īlice: That’s right. And fewer photons means fewer possibilities to arrange them, means less entropy. If each photon has a lot of energy, you need fewer photons to reach the total energy.īob: I see. Since these photons have a rather high energy, they are relatively few and therefore have low entropy.īob: Why are there fewer photons if they have a lot of energy?Īlice: Because the total energy is the number of photons times the energy of a single photon. In that process, some energy (photons) get absorbed. Plants make photosynthesis with this light and split the carbon dioxide (CO2) in the air into carbon (C) and oxygen (O2). The light that arrives from the sun is mainly in the visible range. Where does my food get its low entropy from? Wasn’t it all earth, air and water with high entropy at some point?Īlice: Excellent question! The solution is the sun. 
Does that mean that if I don’t use the energy from my food to order my life, according to the second law of thermodynamics, it automatically gets worse and worse?īob: But I don’t quite understand it yet. Can you apply that to life? There are few ways that my life can go the way I want it to, and many ways that my life falls appart. This means that as you decrease the entropy of the marbles, the entropy in your body increases! Now the second law of thermodynamics says that after you put the marbles in order the total entropy still increases, even though the entropy of the box alone might decrease.īob: Ah okay. So a pile of shit has more entropy than a quinoa-avocado salad. There are fewer ways to arrange atoms to make a quinoa-avocado salad and many ways to make a pile of shit. 
For those calories, your intestines turn vegan quinoa avocado salad into a giant pile of shit. But to sort the marbles, you need energy. But what if I now sort the balls by hand? Then entropy decreases!Īlice: Only if you look at the box in isolation. Sure, what happens is what has the most possibilities. This is the famous second law of thermodynamics.īob: Cool! That’s totally logical. They will always mix, because there are more possibilities for this to happen. If there are many balls, these probabilities are so extremely different that the balls never sort themselves spontaneously by shaking. And since there are many ways that the balls will mix, the probability that this will happen is very high. Since there’s only one way that the marbles can perfectly sort themselves out in color, the probability that this happens by shaking is extremely low. Many possibilities for a certain result means high entropy and few possibilities we call low entropy.Īlice: Right. The entropy of this arrangement is high because there are many possibilities to arrive at a random pattern.īob: I understand. How many possible arrangements of the balls are there to make a complicated pattern of red and blue balls?Īlice: Yes. Now I have a random complicated pattern of blue and red balls, right?Īlice: Correct. What do you think happens?īob: Well, the marbles will mix. Now you shake the box and wait for the marbles to cover the ground again. The entropy is low, in this case even zero. For this arrangement there is only one possibility. How many possibilities are there to arrange the marbles this way assuming that marbles having the same color are identical?Īlice: Right. Now you arrange the marbles so that exactly the right half is blue and the left half is red. What does entropy mean? And why does it never decrease? And what does it have to do with disorder or order? Entropy is simply explained to you here.īob: Hey Alice, what is the definition of entropy? And what does it have to do with physics?Īlice: Entropy is a measure of the number of possibilities.Īlice: Imagine a box whose bottom is completely covered with red and blue marbles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
